
Statistical Tools For Pharmaceuticals
This 4-day track provides participants with the foundation for effectively using statistical methods to validate a pharmaceutical process.
Statistical Tools For Pharmaceuticals
Learn to apply Minitab tools commonly used in the pharmaceutical industry. Develop sound statistical approaches to data analysis by understanding how to select the right tool for a given scenario and to correctly interpret the results of the analysis. Learn how to easily import data and export output.
Learn the foundation for important statistical concepts for determining if a process mean is off target, whether two means are significantly different, and for demonstrating if a process change does not significantly affect a critical response. Develop the necessary skills to successfully evaluate and certify measurement systems. Understand how to utilize important capability analysis tools to evaluate your processes relative to internal and customer specifications. Learn how to evaluate a random sample of product from a lot to determine whether to accept or reject the entire lot.
Understand how to apply DOE for process improvement. Learn how to use stability analysis for determining the shelf life of a product. All applications place emphasis on making good business decisions based upon the practical application of statistical techniques commonly used in the pharmaceutical industry.
Topics include
Bar Charts
Histograms
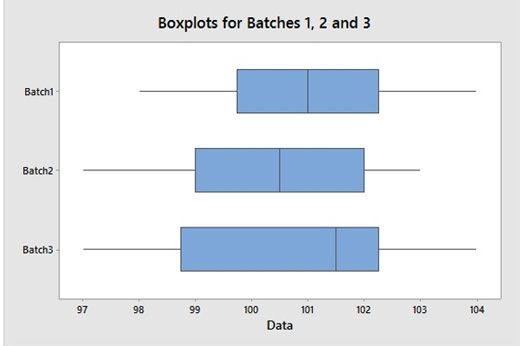
Boxplots
Scatterplots
Power and Sample Size Determination
t-Tests
Equivalence Tests
Proportion Tests
Tolerance Intervals
Regression
One-Way ANOVA
Multi-Variable ANOVA
DOE
Attribute Agreement Analysis & Gage R&R
Attribute Acceptance Sampling
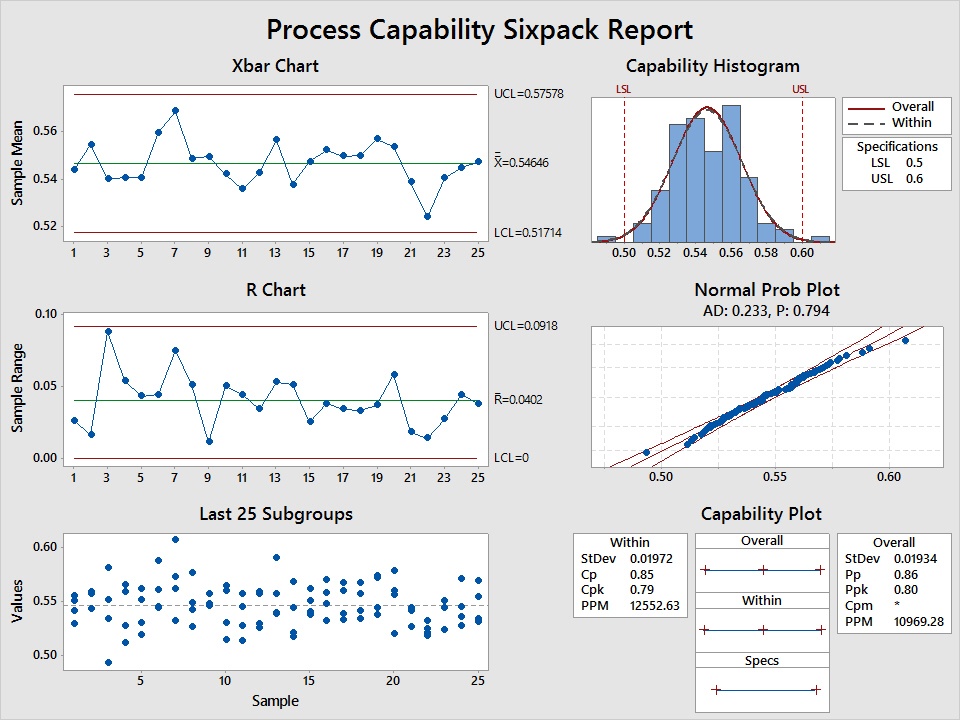
Capability Analysis for Normal and Nonnormal Data
Prerequisite:
On- Site Training
Training for an organization’s employees at its site. Minimum participants required-10 Nos. Training module (Service Quality / Manufacturing), level of the training and date would be scheduled as per mutual convenience.
The benefits of on-site training are:
- Bringing our trainer on-site is the most convenient and cost-effective way to develop the skills your teams need to improve processes.
- No travel required for the participants.
- Customization could be possible within our training scope. Choose from prepared learning tracks or create your own by selecting from our individual course offerings.
Upcoming Live Online Public Training Schedule
Statistical Tools for Pharmaceuticals
- 23rd, 24th, 30th September & 1st October 2024
- Registration ends on 16th September 2024
- 10:00 AM - 06:00 PM (IST)
- Zoom Online
- 25th, 26th, 28th & 29th November 2024
- Registration ends on 18th November 2024
- 10:00 AM - 06:00 PM (IST)
- Zoom Online
- 23rd, 24th, 26th & 27th December 2024
- Registration ends on 16th December 2024
- 10:00 AM - 06:00 PM (IST)
- Zoom Online
IMP Note: For a Group of 10+ Participants, we schedule an exclusive program, on a mutually convenient date & time.
Contact us to know the details.
Classroom Public Training
We offer Training at many convenient locations in India. Sessions are designed for beginners as well as for experienced professional. We schedule date, time, module (Service Quality / Manufacturing) and venue. It remains open to all and we limit enrollment so our trainers have time to address individual’s query.
Attending along with participants from mixed verticals & functions, has rich take-aways.
Upcoming Classroom Public Training Schedule
Exclusive Program Only
Client's Speak: Testimonials
The training session is very good. I learnt about so many statistical tools which can be used in our daily work.
– Somesh Biswal, Alkem Laboratories Ltd
The way the training and the examples designed is very practical and a layman (non-statistician) can understand.
– Suprava Sahoo, Zydus Cadila Healthcare
This program is very useful in terms of improving knowledge about statistics and its applications in pharma industry. This program is having a high potential to tackle and interpret a very large amount of data in very less time.
– Aditya Kumar Singh, Macleods Pharmaceuticals Ltd
The course is designed as per the examples of pharmaceutical application. These practical examples will be used in our professional practice.
– Shailesh Kumar, Intas Pharmaceuticals Ltd
Client's Speak: Watch the video
Qsutra – the Authorised Partner & Certified Training Provider for Minitab in India, Sri Lanka, Bangladesh & Nepal!
With Minitab Certified Trainings, attendees learn to efficiently use Minitab Software and Maximize the Returns, for their organisation’s investments. Our Trainers are Expert Statisticians and Seasoned Industry Professionals, who are tested & certified by Minitab, LLC. USA, after going through their rigorous certification process. Our trainings provide Hands-on learning through real world problem solving – training attendees work through real world quality improvement scenarios (we have standard Examples) with guidance from the instructors so the training is retained after the class ends.